Heavy Metal Based Promotion of Antibiotic Resistance
Awais Paracha
Senior, College of Arts and Sciences
Abstract
Regulation and effective control of microorganism is critical for the preservation of human health especially in the long term. In order to investigate one vector of controlling microorganisms, I investigated the effects of heavy metals on microorganisms. Specifically, this review focuses on antibiotic resistance and production in the presence of heavy metals. My research question for this review was to examine if there is greater antibiotic resistance and production in bacteria because of the presence of heavy metals. I hypothesized that heavy metals will promote antibiotic production and antibiotic resistance in bacteria. This was tested by examining literature in the following ways: 1) the effect of heavy metals, 2) the extent of the effect of heavy metals, and 3) the mechanisms by which these effects are undertaken. In all three of the studies, I came to determine that the argument for greater resistance stood stronger. In regards to the effect of heavy metals greater evidence was found for resistance than inhibition because the results on resistance were much more quantitative. Secondly, I came to realize that the extent of metal based resistance was greater because resistance data was clear, based off the number and types of antibiotics to which it confers resistance. Lastly, resistance mechanisms were also far more defined than inhibition mechanisms. In conclusion, in a day age where microbial resistance is growing rapidly we need to take a cautious approach in how we micromanage microbial environments as how we manage our resources today could have disastrous long-term effects.
Keywords
Heavy Metal based Resistance, Microbial Resistance, Heavy Metal based inhibition, Antibiotic Resistance, Antibiotic Production
Introduction
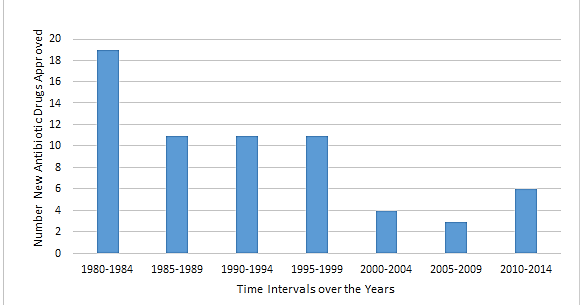
The rising risk of global health via antibiotic resistance remains as critical as ever as nearly 2,000,000 Americans get Hospital Acquired Infections every year with almost 99,000 dying from antibiotic resistant pathogens every year (Diekema et al., 2004). Human health is explicitly at danger because of the rise of various therapeutic failures, as 60% of infectious disease specialists in 2011 had seen a pan-resistant, non-treatable bacterial infection within the last year (Viswanathan, 2014). Additionally, life threatening drug-resistant microbial infections, such as methicillin-resistant Staphylococcus aureus (MRSA) which accounts for 11,285 deaths per year (Gross, 2013) and vancomycin-resistant Enterococci (VRE) which account for 1,300 deaths per year have come to dominate the public health scene (Ventola, 2015). In lieu of all this, there have been increased mortality by about 99,000 deaths due to antibiotic resistant pathogens, extended hospital stays by about 6.4 to 12.7 days, lower number of newly developed antibiotics (Figure 1), and higher costing alternative therapy with the treatment for a patient antibiotic resistant infection costing an extra $18, 588-$29,069 (Ventola, 2015).
Regulation and effective control of microorganism is critical for the preservation of human health especially in the long term. In order to investigate one vector of controlling microorganisms, I investigated the effects of heavy metals on microorganisms. Specifically, this review focuses on antibiotic resistance and production in the presence of heavy metals. My research question for this review was to examine if there is greater antibiotic resistance and production in bacteria because of the presence of heavy metals. I hypothesized that heavy metals will promote antibiotic production and antibiotic resistance in bacteria. This was tested by examining literature in the following ways: 1) the effect of heavy metals, 2) the extent of the effect of heavy metals, and 3) the mechanisms by which these effects are undertaken. In all three of the studies, I came to determine that the argument for greater resistance stood stronger. In regards to the effect of heavy metals greater evidence was found for resistance than inhibition because the results on resistance were much more quantitative. Secondly, I came to realize that the extent of metal based resistance was greater because resistance data was clear, based off the number and types of antibiotics to which it confers resistance. Lastly, resistance mechanisms were also far more defined than inhibition mechanisms. In conclusion, in a day age where microbial resistance is growing rapidly we need to take a cautious approach in how we micromanage microbial environments as how we manage our resources today could have disastrous long-term effects.
Keywords
Heavy Metal based Resistance, Microbial Resistance, Heavy Metal based inhibition, Antibiotic Resistance, Antibiotic Production
Introduction
The rising risk of global health via antibiotic resistance remains as critical as ever as nearly 2,000,000 Americans get Hospital Acquired Infections every year with almost 99,000 dying from antibiotic resistant pathogens every year (Diekema et al., 2004). Human health is explicitly at danger because of the rise of various therapeutic failures, as 60% of infectious disease specialists in 2011 had seen a pan-resistant, non-treatable bacterial infection within the last year (Viswanathan, 2014). Additionally, life threatening drug-resistant microbial infections, such as methicillin-resistant Staphylococcus aureus (MRSA) which accounts for 11,285 deaths per year (Gross, 2013) and vancomycin-resistant Enterococci (VRE) which account for 1,300 deaths per year have come to dominate the public health scene (Ventola, 2015). In lieu of all this, there have been increased mortality by about 99,000 deaths due to antibiotic resistant pathogens, extended hospital stays by about 6.4 to 12.7 days, lower number of newly developed antibiotics (Figure 1), and higher costing alternative therapy with the treatment for a patient antibiotic resistant infection costing an extra $18, 588-$29,069 (Ventola, 2015).
Figure 1: New antibiotic drug approval though the years. Over the last several decades new antibiotic production has consistently dropped. Data courtesy of “The Antibiotic Resistance Crisis” (Ventola, 2015).
In regards to addressing the problem of growing antibiotic resistance and rising superbugs, my review article is focused on antibiotic resistance and production in the presence of heavy metals. My research question for this review was to examine if there is greater antibiotic resistance and production in soil bacteria because of the presence of heavy metals in the soil. Having stated that, based on preliminary studies and published experiments (Hu, Cheng, & Tao,
2017), I hypothesized that heavy metals will promote antibiotic production and antibiotic resistance in soil bacteria.
My hypothesis which supposes that heavy metals will promote antibiotic resistance and antibiotic production in soil bacteria will be tested in the following ways: 1) the effect of heavy metals, 2) the extent of the effect of heavy metals, and 3) the mechanisms by which these effects are undertaken. In all three of the studies, I will first determine if heavy metals promote or inhibit antibiotic resistance and production.
As ever, there remains a persistent threat of antibiotic resistance in common bacteria such as S. aureus (Fischbach & Walsh, 2009), Enterococcus faecium (Fisher & Phillips, 2009), and Streptococcus pneumoniae (Zinner, 2005). Combine that threat with the widespread abuse and prevalence in the prescription of antibiotics (Adegoke, Faleye, Singh, & Stenstrom, 2016), the threat of antibiotic-resistant bacteria becoming a legitimate health threat is as real as can be. Therefore, the only way to win this race of biological arms is to stay one step ahead of resistant bacteria by investigating any supplemental vectors, such as heavy metals, that may alter antibiotic production and resistance in soil bacteria. Thus the goal of this project was to investigate the potential greater antibiotic resistance and production by bacteria that are in heavy metal settings as compared to bacteria that are not exposed to heavy metals in large quantities (Brussow, 2017).
Determination of the effect of Heavy Metals on Bacterial Antimicrobial Production and Resistance
Heavy Metal based promotion of bacterial antimicrobial production and resistance
Whilst investigating the effect of heavy metals it is important to see if antimicrobial production and resistance rise because of exposure to heavy metals and, if they do generally via what scale and mechanism do they carry out these changes. This is very critical because of the large amount of heavy metals that have been shown to be present in natural environments and because of heavy commerical use (Yousefi et al., 2017).
Specfically, in the case of natural environments a specific example of this would the be the use of copper and zinc by protozoa to kill the bacteria after the “Great Oxidation Event” (Poole, 2017). Briefly, after molecular oxygen appeared, natural selection promoted the use of copper by organisms such as protoza to reduce oxygen. Due to this rapid need and use for copper, protozoa would go onto rapidly incorporate this into their predatory mechanisms for consuming bacteria. Thus, because of this predatory mechanism we had a greater selection for copper resistance in bacteria (Hao et al., 2016). Thus the fact that protozoa used copper as a way to poison and then kill their prey this selective evoloution pressure could have led to the development of copper resistance. This evoloutionary development can be further suppourted by the observation that copper resistance is present across almost all microbial environments. Thus, it would be fair to propose that the rise in resistance could be very well attributed to the predatory mechanisms used by early protozoans (German, Doyscher, & Rensing, 2013).
Additionally several studies (Zhu et al., 2013) were conducted on the agriculutral uses of various fertilizers and those shown to have metals present tended to have a correlation with a higher presence of antibiotic-resistant genes (Silver & Phung, 1996). Thus, in these cases a greater presence of antibiotic-resistant genes would contribute to greater antibiotic resistance (Lin et al., 2016). Another clear showcase of the rise in antibiotic-resistance can be due to the link present between antibiotic-resistance and heavy metal-resistant genes (Davison, 1999). Thus, because these can coexist in the same genetic element we can permit for antimicrobial resistance selection to be heavily promoted in environemts that do have a heavy presence of these heavy metals (Alonso, Sanchez, & Martinez, 2001). Thus by viewing the greater presence of antibiotic resistant genes in soil additionally suppemented by the presence of heavy metal resistnat genes it is very important to realize that this may correlate with the presence of heavy metals in these environments.
Correlations between antibiotic-resistance and heavy metals have also been observed in the clinical realm in regards to the treatment of burns (Hobman & Crossman, 2015). Burns are usually treated by silver nitrate (Norman et al., 2017). However, because of the extensive use of silver nitrate in burn wounds, burn wounds tend to be easily infrected by pathogens (Pruitt, McManus, Kim, & Goodwin, 1998). This was classic example of the genetic linkage in the coselection between silver and antibiotic resistance (McHugh, Moellering, Hopkins, & Swartz, 1975). Hence, we can see that by looking at clinical cases especially of burn cases there is a clear rise in genetic resistance because of the presence of a genetic linkage in the coselection between certain metals and antibiotic resistance.
Lastly, studies were done in which control soils were compared with soils that contained heavy metals namely mercury, and clearly the soil contained a greater amount of antibioitic-resistant strains as compared to the control soil as witnessed by the lower amount of plasmids contained in the control group samples (Rasmussen & Sorensen, 1998). This presence of greater resistance plasmids in heavy metal environments (Amin, Latif, & Opella, 2017) is something that should definetly be considered of extreme importance as it something that is present not only in soil environments but other various environents such as wastewater or polluted environments.
Thus, in regards to resistance it can be seen that there is greater resistance because of the presence of heavy metals in natural predatory mechanisms, greater antibiotic resistant genes in heavy metal environments, correlation between antibiotic resistance and heavy metal resistant genes, and plasmid amount in regards to environment metal concentration. Thus, by looking at all these facets of microbiological environments we can believe that the presence of heavy metals promotes the idea of greater antibiotic resistance.
Heavy Metal based inhibition of bacterial antimicrobial production and resistance
On the other hand, whilst investigating the effect of heavy metals it is important to see if antimicrobial production and resistance decrease because of exposure to heavy metals and if they do generally via what scale and mechanism. I think various factors need to be considered for the presence of heavy metals and their toxicity to bacteria which in essence leads to resistance.
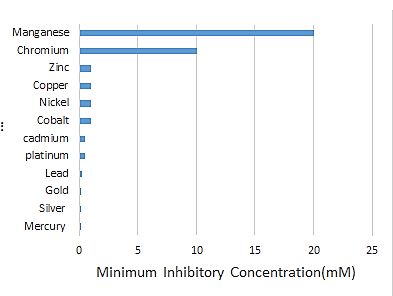
First, metals like zinc, nickel, chromium, copper, and cobalt do hold important functions in the phyisiology of the cell, so they may not hold as much importance in the realm of toxicity (Nies, 1999). In essence, certain metal ions are required in several cellular process as parts of metalloproteins and they serve in the role of a cofactor or structural element for an enzyme. However, it is essential for bacteria to regulate proper metal uptake and make sure availability is in accordance with physiological needs, as an imbalance in bacterial metal homeostasis can be extremely harmful to the bacteria (Porcheron, Garenaux, Proulx, Sabri, & Dozois, 2013). Because of this, I think it is very important to realize that the inhibitory or promotory effects of these metals depend on their concentration. In order to delineate, the minimum inhibitory concentration (MIC) of various heavy metals, is the lowest concentration of a chemical, substance, or element that averts bacterial growth. In essence, the more deadly the metal is the lower it is MIC will be because a lower concentration will be needed to cause an inhibitory response. For example, in E. coli, the MIC of manganese is 20 mM, while it is 10 mM for a metal like chromium, thus chromium is twice as toxic than manganese (Figure 2).
In regards to addressing the problem of growing antibiotic resistance and rising superbugs, my review article is focused on antibiotic resistance and production in the presence of heavy metals. My research question for this review was to examine if there is greater antibiotic resistance and production in soil bacteria because of the presence of heavy metals in the soil. Having stated that, based on preliminary studies and published experiments (Hu, Cheng, & Tao,
2017), I hypothesized that heavy metals will promote antibiotic production and antibiotic resistance in soil bacteria.
My hypothesis which supposes that heavy metals will promote antibiotic resistance and antibiotic production in soil bacteria will be tested in the following ways: 1) the effect of heavy metals, 2) the extent of the effect of heavy metals, and 3) the mechanisms by which these effects are undertaken. In all three of the studies, I will first determine if heavy metals promote or inhibit antibiotic resistance and production.
As ever, there remains a persistent threat of antibiotic resistance in common bacteria such as S. aureus (Fischbach & Walsh, 2009), Enterococcus faecium (Fisher & Phillips, 2009), and Streptococcus pneumoniae (Zinner, 2005). Combine that threat with the widespread abuse and prevalence in the prescription of antibiotics (Adegoke, Faleye, Singh, & Stenstrom, 2016), the threat of antibiotic-resistant bacteria becoming a legitimate health threat is as real as can be. Therefore, the only way to win this race of biological arms is to stay one step ahead of resistant bacteria by investigating any supplemental vectors, such as heavy metals, that may alter antibiotic production and resistance in soil bacteria. Thus the goal of this project was to investigate the potential greater antibiotic resistance and production by bacteria that are in heavy metal settings as compared to bacteria that are not exposed to heavy metals in large quantities (Brussow, 2017).
Determination of the effect of Heavy Metals on Bacterial Antimicrobial Production and Resistance
Heavy Metal based promotion of bacterial antimicrobial production and resistance
Whilst investigating the effect of heavy metals it is important to see if antimicrobial production and resistance rise because of exposure to heavy metals and, if they do generally via what scale and mechanism do they carry out these changes. This is very critical because of the large amount of heavy metals that have been shown to be present in natural environments and because of heavy commerical use (Yousefi et al., 2017).
Specfically, in the case of natural environments a specific example of this would the be the use of copper and zinc by protozoa to kill the bacteria after the “Great Oxidation Event” (Poole, 2017). Briefly, after molecular oxygen appeared, natural selection promoted the use of copper by organisms such as protoza to reduce oxygen. Due to this rapid need and use for copper, protozoa would go onto rapidly incorporate this into their predatory mechanisms for consuming bacteria. Thus, because of this predatory mechanism we had a greater selection for copper resistance in bacteria (Hao et al., 2016). Thus the fact that protozoa used copper as a way to poison and then kill their prey this selective evoloution pressure could have led to the development of copper resistance. This evoloutionary development can be further suppourted by the observation that copper resistance is present across almost all microbial environments. Thus, it would be fair to propose that the rise in resistance could be very well attributed to the predatory mechanisms used by early protozoans (German, Doyscher, & Rensing, 2013).
Additionally several studies (Zhu et al., 2013) were conducted on the agriculutral uses of various fertilizers and those shown to have metals present tended to have a correlation with a higher presence of antibiotic-resistant genes (Silver & Phung, 1996). Thus, in these cases a greater presence of antibiotic-resistant genes would contribute to greater antibiotic resistance (Lin et al., 2016). Another clear showcase of the rise in antibiotic-resistance can be due to the link present between antibiotic-resistance and heavy metal-resistant genes (Davison, 1999). Thus, because these can coexist in the same genetic element we can permit for antimicrobial resistance selection to be heavily promoted in environemts that do have a heavy presence of these heavy metals (Alonso, Sanchez, & Martinez, 2001). Thus by viewing the greater presence of antibiotic resistant genes in soil additionally suppemented by the presence of heavy metal resistnat genes it is very important to realize that this may correlate with the presence of heavy metals in these environments.
Correlations between antibiotic-resistance and heavy metals have also been observed in the clinical realm in regards to the treatment of burns (Hobman & Crossman, 2015). Burns are usually treated by silver nitrate (Norman et al., 2017). However, because of the extensive use of silver nitrate in burn wounds, burn wounds tend to be easily infrected by pathogens (Pruitt, McManus, Kim, & Goodwin, 1998). This was classic example of the genetic linkage in the coselection between silver and antibiotic resistance (McHugh, Moellering, Hopkins, & Swartz, 1975). Hence, we can see that by looking at clinical cases especially of burn cases there is a clear rise in genetic resistance because of the presence of a genetic linkage in the coselection between certain metals and antibiotic resistance.
Lastly, studies were done in which control soils were compared with soils that contained heavy metals namely mercury, and clearly the soil contained a greater amount of antibioitic-resistant strains as compared to the control soil as witnessed by the lower amount of plasmids contained in the control group samples (Rasmussen & Sorensen, 1998). This presence of greater resistance plasmids in heavy metal environments (Amin, Latif, & Opella, 2017) is something that should definetly be considered of extreme importance as it something that is present not only in soil environments but other various environents such as wastewater or polluted environments.
Thus, in regards to resistance it can be seen that there is greater resistance because of the presence of heavy metals in natural predatory mechanisms, greater antibiotic resistant genes in heavy metal environments, correlation between antibiotic resistance and heavy metal resistant genes, and plasmid amount in regards to environment metal concentration. Thus, by looking at all these facets of microbiological environments we can believe that the presence of heavy metals promotes the idea of greater antibiotic resistance.
Heavy Metal based inhibition of bacterial antimicrobial production and resistance
On the other hand, whilst investigating the effect of heavy metals it is important to see if antimicrobial production and resistance decrease because of exposure to heavy metals and if they do generally via what scale and mechanism. I think various factors need to be considered for the presence of heavy metals and their toxicity to bacteria which in essence leads to resistance.
First, metals like zinc, nickel, chromium, copper, and cobalt do hold important functions in the phyisiology of the cell, so they may not hold as much importance in the realm of toxicity (Nies, 1999). In essence, certain metal ions are required in several cellular process as parts of metalloproteins and they serve in the role of a cofactor or structural element for an enzyme. However, it is essential for bacteria to regulate proper metal uptake and make sure availability is in accordance with physiological needs, as an imbalance in bacterial metal homeostasis can be extremely harmful to the bacteria (Porcheron, Garenaux, Proulx, Sabri, & Dozois, 2013). Because of this, I think it is very important to realize that the inhibitory or promotory effects of these metals depend on their concentration. In order to delineate, the minimum inhibitory concentration (MIC) of various heavy metals, is the lowest concentration of a chemical, substance, or element that averts bacterial growth. In essence, the more deadly the metal is the lower it is MIC will be because a lower concentration will be needed to cause an inhibitory response. For example, in E. coli, the MIC of manganese is 20 mM, while it is 10 mM for a metal like chromium, thus chromium is twice as toxic than manganese (Figure 2).
Figure 2: Minimum Inhibitory Concentration (MIC) values for specific heavy metals for E. coli. In this figure, heavy metal toxicity in E. coli is compared by assessing the minimum inhibitory concentration (MIC) of various heavy metals. The MIC value is the lowest concentration of a chemical, substance, or element that prevents bacterial growth. Thus, the more toxic the metal the lower it is MIC will be because a smaller concentration will be needed to provoke an inhibitory response amongst the bacteria. Data courtesy of “Microbial Heavy-Metal Resistance Crisis” (Nies, 1999).
The level of inhibition of antimicrobial production and resistance is also highly dependent on the type of metal (Seiler & Berendonk, 2012). For example, Pseudomonas aeruginosa has a metal toxicity ranking in which it is more sensative to lead then copper and then zinc (Table 1). In summary, it is very important to consider all these factors in metal toxicity as it pertains to decreased antibiotic resistance.
Table 1*: Metal Toxicity Ranking
The level of inhibition of antimicrobial production and resistance is also highly dependent on the type of metal (Seiler & Berendonk, 2012). For example, Pseudomonas aeruginosa has a metal toxicity ranking in which it is more sensative to lead then copper and then zinc (Table 1). In summary, it is very important to consider all these factors in metal toxicity as it pertains to decreased antibiotic resistance.
Table 1*: Metal Toxicity Ranking
*Table is from “Heavy metal driven co-selection of antibiotic resistance in soil and water bodies impacted by agriculture and aquaculture” (Seiler & Berendonk, 2012).
Additionally, I also think that the toxicity of these metals can depend on other abiotic factors such as pH (Schulz-Zunkel & Krueger, 2009), the concentration of organic matter (Nicholson, Smith, Alloway, Carlton-Smith, & Chambers, 2003) and the redox potential (Mergeay et al., 1985). For example, if there is high concentration of organic matter, zinc and copper solubility go down so there toxicity goes down (Schulz-Zunkel & Krueger, 2009).
Finally, there are also certain other factors that go into determining if a metal can prove to be toxic to bacteria, this toxicity is also impacted by the type of bacteria as Gram-positive bacteria are more sensitive than Gram-negative bacteria (Sterritt & Lester, 1980). In a study on sewage samples in the presence of the heavy metal chromium, the following bacteria were looked at: E. coli, P. aeruginosa, Klebsiella pneumoniae, S. aureus and Enterococcus faecalis. In this study, when evaluating bacterial composition, 2 different trials were run at two independent locations, and all of the results showed an overwhelmingly increase in Gram-positive bacteria present in the bacterial samples (Lima de Silva et al., 2012). Clearly, Gram-positive bacteria are more sensitive to the presence of heavy metals than Gram-negative bacteria.
Thus, in regards to resistance it can be seen that there is lower resistance if we consider it based on toxicity because of the presence of certain metals, metal concentration, pH value, concentration of organic matter, redox potential, and bacteria type. Thus, by looking at all these aspects or features of microbiological environments along with bacterial properties we can believe that the presence of heavy metals can in certain scenarios promote the idea of lower antibiotic resistance.
Determination of the extent of Heavy Metals on Bacterial Antimicrobial Production and Resistance
Extent of Heavy Metal based promotion of bacterial antimicrobial production and resistance
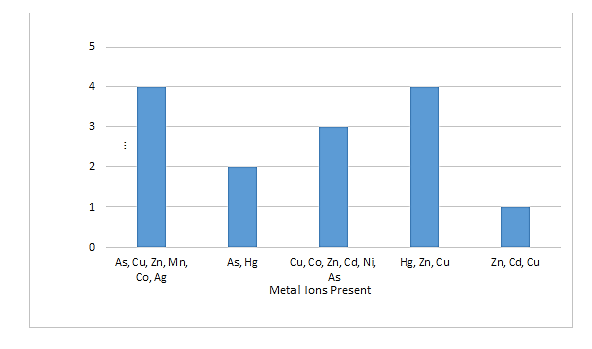
The presence of metals does have a large potential impact on resistance. This seems to be a prominent concern because these metals are long lasting and can confer indirect resistance. In this section, we will look at the extent of resistance by comparing metal ion presence to co-selection against certain antibiotics (Figure 3). Different metal ions confer resistance in different fashions or mechanisms. It has been shown that metal ions including As, Cu, Zn, Mn, Co, and Ag work by reducing membrane permeability to antibiotics thus by doing this the bacteria mentioned reduced their susceptibility to the following antibiotics, ciprofloxacin, tetracycline, chloramphenicol, and β-lactams(Silver & Phung, 1996). Certain other heavy metals function by altering cellular targets these include Hg, Zn, and Cu. By altering cellular targets they confer resistance to ciprofloxacin, rifampicin, trimethoprim, and β-lactams (Barkay, Miller, & Summers, 2003). Specifically, Co, Cu, Zn, Cd, Ni, and As function to confer resistance of tetracycline, chloramphenicol, and β-lactams by promoting rapid efflux of the antibiotic (Nies, 2003). Metals like Hg and As inactivate the drug or antibiotic and these consequently result in resistance to β-lactams and chloramphenicol (Mukhopadhyay & Rosen, 2002). Lastly, metals like Zn, Cd, and Cu function via drug sequestering, as in the case of the antibiotic coumermycin (Bontidean et al., 2000). Thus, we can measure the extent to which a metal or metals confer resistance by looking at the number and type of antibiotics to which it confers resistance.
Additionally, I also think that the toxicity of these metals can depend on other abiotic factors such as pH (Schulz-Zunkel & Krueger, 2009), the concentration of organic matter (Nicholson, Smith, Alloway, Carlton-Smith, & Chambers, 2003) and the redox potential (Mergeay et al., 1985). For example, if there is high concentration of organic matter, zinc and copper solubility go down so there toxicity goes down (Schulz-Zunkel & Krueger, 2009).
Finally, there are also certain other factors that go into determining if a metal can prove to be toxic to bacteria, this toxicity is also impacted by the type of bacteria as Gram-positive bacteria are more sensitive than Gram-negative bacteria (Sterritt & Lester, 1980). In a study on sewage samples in the presence of the heavy metal chromium, the following bacteria were looked at: E. coli, P. aeruginosa, Klebsiella pneumoniae, S. aureus and Enterococcus faecalis. In this study, when evaluating bacterial composition, 2 different trials were run at two independent locations, and all of the results showed an overwhelmingly increase in Gram-positive bacteria present in the bacterial samples (Lima de Silva et al., 2012). Clearly, Gram-positive bacteria are more sensitive to the presence of heavy metals than Gram-negative bacteria.
Thus, in regards to resistance it can be seen that there is lower resistance if we consider it based on toxicity because of the presence of certain metals, metal concentration, pH value, concentration of organic matter, redox potential, and bacteria type. Thus, by looking at all these aspects or features of microbiological environments along with bacterial properties we can believe that the presence of heavy metals can in certain scenarios promote the idea of lower antibiotic resistance.
Determination of the extent of Heavy Metals on Bacterial Antimicrobial Production and Resistance
Extent of Heavy Metal based promotion of bacterial antimicrobial production and resistance
The presence of metals does have a large potential impact on resistance. This seems to be a prominent concern because these metals are long lasting and can confer indirect resistance. In this section, we will look at the extent of resistance by comparing metal ion presence to co-selection against certain antibiotics (Figure 3). Different metal ions confer resistance in different fashions or mechanisms. It has been shown that metal ions including As, Cu, Zn, Mn, Co, and Ag work by reducing membrane permeability to antibiotics thus by doing this the bacteria mentioned reduced their susceptibility to the following antibiotics, ciprofloxacin, tetracycline, chloramphenicol, and β-lactams(Silver & Phung, 1996). Certain other heavy metals function by altering cellular targets these include Hg, Zn, and Cu. By altering cellular targets they confer resistance to ciprofloxacin, rifampicin, trimethoprim, and β-lactams (Barkay, Miller, & Summers, 2003). Specifically, Co, Cu, Zn, Cd, Ni, and As function to confer resistance of tetracycline, chloramphenicol, and β-lactams by promoting rapid efflux of the antibiotic (Nies, 2003). Metals like Hg and As inactivate the drug or antibiotic and these consequently result in resistance to β-lactams and chloramphenicol (Mukhopadhyay & Rosen, 2002). Lastly, metals like Zn, Cd, and Cu function via drug sequestering, as in the case of the antibiotic coumermycin (Bontidean et al., 2000). Thus, we can measure the extent to which a metal or metals confer resistance by looking at the number and type of antibiotics to which it confers resistance.
Figure 3: Number of antibiotics resistant in regards to metal ions present. Certain metal ions promote resistance to a greater number of antibiotics as compared to others in prokaryotic systems. Data courtesy of “Co-selection of antibiotic and metal resistance” (Baker-Austin, Wright, Stepanauskas, & McArthur, 2006).
Extent of Heavy Metal based inhibition of bacterial antimicrobial production and resistance
Interestingly, an argument can be made for the other side, too. The presence of metals does have potential impact on lowering antibiotic resistance. For example, in a study done on several metal contaminated streams in the United States, a surprising trend was found that a greater metal concentration correlated with a lower amount of resistance, thus showing a negative correlation between antibiotic resistance and microbial production (Tuckfield &McArthur, 2008). Interestingly, another study examining the levels on antibiotic resistance in pig manure would go on to further our beliefs on the extent of decreased antimicrobial resistance in the presence of heavy metals (Stepanauskas et al., 2005) In that study, lead was shown to have a negative correlation with antibiotic resistance in response to ampicillin, amoxicillin+ clavulanate, and piperacillin, while mercury was shown to have a negative correlation with antibiotic resistance in response to the those antibiotics along with chloramphenicol, doxycycline, neomycin, spectinomycin, and streptomycin (Holzel et al., 2012). Thus, by looking at the correlations between antibiotic resistance and heavy metals we can specifically gauge the extent to which this decrease is significant and be able to identify the extent to which resistance has decreased in the presence of heavy metals.
Determination of the mechanism by which heavy metals alter Antimicrobial Production and Resistance
Mechanisms of heavy metal based promotion of bacterial antimicrobial production and resistance
It is important to determine not the only the degree of increased resistance but it is very important to acknowledge the role of mechanisms by which this occurs. Overall there are three very important mechanisms which have bacteria have evolved to avoid cellular damage that occurs because of the presence of metal ions.
The first mechanism would be sequestering or forming complexes with toxic metals. As soon as the metal binds to the bacterium, the toxic metal ion concentrations of metal ions are very rapidly diminished. These toxic concentrations are diminished through rapid sequesteration which isolates metals prevent them from causing damage or inhabiting pathways. Moreover, cell membranes, cell walls, and other cell structures have been known to conduct biosorption of toxic metals (Harrison, Ceri, & Turner, 2007). In a study on S. aureus, a bacterium that uses calprotectin (the most common protein in the cytoplasm of neutrophils), calprotectin was able to increase virulence and mortalilty in the host through the sequestering of zinc and manganese ions, which then promotes the activation of certain pathways which encode virulence factors (Cho et al., 2015). Metal buffering permits for the expression of certain proteins to allow for the adaptation in case of extra metals, amongst these proteins are those for the sequestering metals. For example, certain prokaryotes store iron in ferritin and ferritin-related proteins in case of non-normal iron levels (Chandrangsu, Rensing, & Helmann, 2017). Thus in this case, prokaryotes increase resistance by being able to maintain a stable environment which will promote normal homeostatic functions. Clearly, sequestering is an important technique that various different types of prokaryotes make use of in order to promote resistance.
A second very important mechanism of toxic metal resistance is via detoxification via intracellular ion reduction. By reducing a heavy metal ion it will be put in an oxidation state where it can diffuse out of the cell and thus not be toxic to it anymore. This can be shown by the example of merA gene which encode mercury reductase which reduces mercury2+ to mercury0, then allowing this form of mercury to diffuse out because of low point of evaporation (Schiering et al., 1991). In another study this was exhibited by Enterobacter cloacae, which gained resistance to arsenate through reduction of arsenate with the help of bacterium-produced arsenate reductase (Chen et al., 2015). Certain microbes can use heavy metals like iron and metal as the only electron acceptor in the oxidation of organic compounds. For example, by simultaneouslyoxidzaing an organic compound while using a metal like Fe(III) or Mn(IV), microbes can generate energy (Lovley & Phillips, 1988) . Thus, in this case microbes indirectly increase resistance by being able to conduct metabolic processes at a higher level. Through perusing the literature it becomes evident that reduction is a key vector for inducing resistance in prokaryotes.
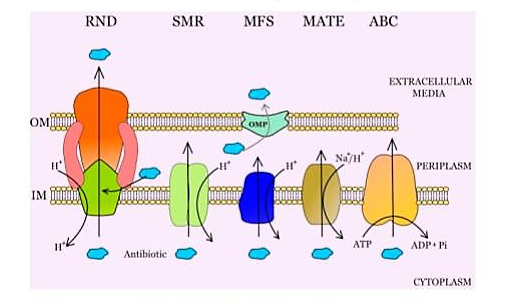
A third mechanism would take care of toxic ions via efflux systems. Efflux systems are active transporters that pump toxins out of the cell (Blanco et al., 2016). In essence, there are 6 major efflux superfamilies which include: 1) the small multidrug resistance family(Handzlik, Matys, & Kiec-Kononowicz, 2013). 2) the resistance- nodulation-division family (Alvarez-Ortega, Olivares, & Martínez, 2013), 3) the adenosine tripohosphate binding cassette superfamily (Lubelski, Konings, & Driessen, 2007), 4) the major facilitator superfamily(Pao, Paulsen, & Saier, 1998), and 5) the multidrug and toxic compound extrusion superfamily (Lu, 2016)(Figure 4). An example of this would be the Czc antiporter(Resistance Nodulation division superfamily) from Alcaligenes eutrophus, which mediates resistance to certain metal ions by metal efflux from the cytoplasm to the surrounding environment (Silver & Phung, 1996). In another study on heavy metal based pollution it was seen that heavy metal ion based efflux systems decreased tolernace toward antibiotics. Under stress from Cr2O−7 and Cu2+, the SoxS protien was upregulated, and this promotes the AcrAB efflux system (Resistance Nodulation division superfamily) which promotes resistance to antibiotics like chloramphenicol, tetracyclin, novobiocin, nafcillin, and oxacillin (Harrison et al., 2009). Lastly,bacteria often use efflux pumps not only as a means of conferring direct resistance but increasing indirect resistance by helping in the maintainence of cellular levels of ions, many of these ions are often enzmyatic cofactors (Delmar, Su, & Yu, 2014). Thus, metal ion efflux systems play a very large role in conferring antimicrobial resistance whether that be directly or indirectly.
Extent of Heavy Metal based inhibition of bacterial antimicrobial production and resistance
Interestingly, an argument can be made for the other side, too. The presence of metals does have potential impact on lowering antibiotic resistance. For example, in a study done on several metal contaminated streams in the United States, a surprising trend was found that a greater metal concentration correlated with a lower amount of resistance, thus showing a negative correlation between antibiotic resistance and microbial production (Tuckfield &McArthur, 2008). Interestingly, another study examining the levels on antibiotic resistance in pig manure would go on to further our beliefs on the extent of decreased antimicrobial resistance in the presence of heavy metals (Stepanauskas et al., 2005) In that study, lead was shown to have a negative correlation with antibiotic resistance in response to ampicillin, amoxicillin+ clavulanate, and piperacillin, while mercury was shown to have a negative correlation with antibiotic resistance in response to the those antibiotics along with chloramphenicol, doxycycline, neomycin, spectinomycin, and streptomycin (Holzel et al., 2012). Thus, by looking at the correlations between antibiotic resistance and heavy metals we can specifically gauge the extent to which this decrease is significant and be able to identify the extent to which resistance has decreased in the presence of heavy metals.
Determination of the mechanism by which heavy metals alter Antimicrobial Production and Resistance
Mechanisms of heavy metal based promotion of bacterial antimicrobial production and resistance
It is important to determine not the only the degree of increased resistance but it is very important to acknowledge the role of mechanisms by which this occurs. Overall there are three very important mechanisms which have bacteria have evolved to avoid cellular damage that occurs because of the presence of metal ions.
The first mechanism would be sequestering or forming complexes with toxic metals. As soon as the metal binds to the bacterium, the toxic metal ion concentrations of metal ions are very rapidly diminished. These toxic concentrations are diminished through rapid sequesteration which isolates metals prevent them from causing damage or inhabiting pathways. Moreover, cell membranes, cell walls, and other cell structures have been known to conduct biosorption of toxic metals (Harrison, Ceri, & Turner, 2007). In a study on S. aureus, a bacterium that uses calprotectin (the most common protein in the cytoplasm of neutrophils), calprotectin was able to increase virulence and mortalilty in the host through the sequestering of zinc and manganese ions, which then promotes the activation of certain pathways which encode virulence factors (Cho et al., 2015). Metal buffering permits for the expression of certain proteins to allow for the adaptation in case of extra metals, amongst these proteins are those for the sequestering metals. For example, certain prokaryotes store iron in ferritin and ferritin-related proteins in case of non-normal iron levels (Chandrangsu, Rensing, & Helmann, 2017). Thus in this case, prokaryotes increase resistance by being able to maintain a stable environment which will promote normal homeostatic functions. Clearly, sequestering is an important technique that various different types of prokaryotes make use of in order to promote resistance.
A second very important mechanism of toxic metal resistance is via detoxification via intracellular ion reduction. By reducing a heavy metal ion it will be put in an oxidation state where it can diffuse out of the cell and thus not be toxic to it anymore. This can be shown by the example of merA gene which encode mercury reductase which reduces mercury2+ to mercury0, then allowing this form of mercury to diffuse out because of low point of evaporation (Schiering et al., 1991). In another study this was exhibited by Enterobacter cloacae, which gained resistance to arsenate through reduction of arsenate with the help of bacterium-produced arsenate reductase (Chen et al., 2015). Certain microbes can use heavy metals like iron and metal as the only electron acceptor in the oxidation of organic compounds. For example, by simultaneouslyoxidzaing an organic compound while using a metal like Fe(III) or Mn(IV), microbes can generate energy (Lovley & Phillips, 1988) . Thus, in this case microbes indirectly increase resistance by being able to conduct metabolic processes at a higher level. Through perusing the literature it becomes evident that reduction is a key vector for inducing resistance in prokaryotes.
A third mechanism would take care of toxic ions via efflux systems. Efflux systems are active transporters that pump toxins out of the cell (Blanco et al., 2016). In essence, there are 6 major efflux superfamilies which include: 1) the small multidrug resistance family(Handzlik, Matys, & Kiec-Kononowicz, 2013). 2) the resistance- nodulation-division family (Alvarez-Ortega, Olivares, & Martínez, 2013), 3) the adenosine tripohosphate binding cassette superfamily (Lubelski, Konings, & Driessen, 2007), 4) the major facilitator superfamily(Pao, Paulsen, & Saier, 1998), and 5) the multidrug and toxic compound extrusion superfamily (Lu, 2016)(Figure 4). An example of this would be the Czc antiporter(Resistance Nodulation division superfamily) from Alcaligenes eutrophus, which mediates resistance to certain metal ions by metal efflux from the cytoplasm to the surrounding environment (Silver & Phung, 1996). In another study on heavy metal based pollution it was seen that heavy metal ion based efflux systems decreased tolernace toward antibiotics. Under stress from Cr2O−7 and Cu2+, the SoxS protien was upregulated, and this promotes the AcrAB efflux system (Resistance Nodulation division superfamily) which promotes resistance to antibiotics like chloramphenicol, tetracyclin, novobiocin, nafcillin, and oxacillin (Harrison et al., 2009). Lastly,bacteria often use efflux pumps not only as a means of conferring direct resistance but increasing indirect resistance by helping in the maintainence of cellular levels of ions, many of these ions are often enzmyatic cofactors (Delmar, Su, & Yu, 2014). Thus, metal ion efflux systems play a very large role in conferring antimicrobial resistance whether that be directly or indirectly.
Figure 4: Main types of Bacterial Efflux Systems. This is a pictorial representation of the 5 major efflux transporters families: the resistance- nodulation-division family(RND superfamily), the small multidrug resistance family( SMR superfamily), the major facilitator superfamily( MFS Superfamily), the multidrug and toxic compound extrusion superfamily(MATE superfamily), and the adenosine triphosphate binding cassette superfamily(AB
Data courtesy of “Bacterial Multidrug Efflux Pumps: Much More Than Antibiotic Resistance Determinants ” (Blanco et al., 2016).
Although realizing that heavy metals promote resistance, I think it is more important to learn the mechanims by which they confer this resistance as that will help us realize the true essence of heavy metal based resistance. In summary, after looking through the literature on heavy metal based resistance, I found that three of the more critical mechanisms of resistance would include efflux systems, sequesteration, and reduction.
Mechanisms of Heavy Metal based inhibition of bacterial antimicrobial production and resistance
On the contrary, it is important to determine not the only the degree of decreased resistance but it is very important to acknowledge the role of mechanisms by which this occurs. Overall there are three very important mechanisms by which prokaryotes confer decreased resistance.
The first mechanism would be heavy metal based interactions with biochemical sites. In essence, metals like Cd, Hg, and Pb have a quite strong affinity for certain ligands like phosphates, histidyl, and cysteinyl side chains of proteins. By being able to act on such a wide variety of biochemical sites, these metals can inhibit a large variety of enzymes which may include antibiotic degrading enzymes (Vallee & Ulmer, 1972). Another study found that metals like copper can generate hydroxyl radicals while shifting from the reduced to the oxidized state, and these hydroxyl radicals can lead to nucleic acid, protein, and lipid damage (Festa & Thiele, 2011). Moreover, while looking at a study on the prokaryotic topoisomerase, gyrase, it was found that in the presence of calcium ions, gyrase was unable to conduct DNA cleavage at the rate of an enzyme that was not altered by the presence of calcium ions (Sissi & Palumbo, 2009). Clearly, metal ions play a very significant role in their alteration of biochemical pathways via their interaction with certain enzymes or other biomolecules.
The second mechanism would be heavy metal based interactions with antibiotics. Heavy metals can increase the bacteria is exposure to antibiotics by allowing certain proteins to bind them easier. In a study looking at S. aureus mercury and how it localized penicillin binding proteins which would thus allow certain antibiotics like penicillin to be able to bind it in an easier fashion (Paul et al., 1995). Thus, by letting antibiotics bind in an easier fashion, there is a decrease in resistance. In a study on Caulobacter crescentus, the protein StpX, a stalk-specific protein, responded to the presence of copper and zinc and this allowed for the identification and presence of penicillin-binding protein PbpC (Hughes et al., 2013). Therefore, by having metals that localize certain proteins that themselves localize antibiotic binding proteins or are antibiotic binding proteins, we can increase the effectivity of the antibiotic because it binds better resulting in better exposure and a decrease in overall resistance.
Lastly, this decrease in resistance could because of the co-toxic effects. This could mean that a heavy metal happens to degrade or inhibit a protein for antibiotic resistance that bacteria tend to use. Clearly, heavy metals play a large role in biochemical pathways, and have been shown to interact with certain ligands (Vallee & Ulmer, 1972). Moreover, byproducts of certain metal metabolism pathways may also interfere with proteins (Festa & Thiele, 2011). For example, mercury-inhibited enzymes might not regenerate after antibiotic based inhibition of protein synthesis (Holzel et al., 2012). By understanding these commonalities, I think we can understand how a decrease in resistance takes place because of co-toxic effects.
Although realizing that heavy metal mitigate resistance, I think it is more important to learn the mechanims by which they confer this decrease in resistance as that will help us realize the true essence of heavy metal inhibited resistance. In summary, after looking through the literature on heavy metal based resistance, I found that three of the more critical mechanisms of decreased resistance would include heavy metal interference with biochemical pathways, interaction with antibiotics, and co-toxicity.
Conclusion
The objective of this study was to engage in an extensive study on the antibiotic resistance or inhibition in regards in the presence of heavy metals. Specifically, my research question for this review was to examine if there is greater antibiotic resistance and production in soil bacteria because of the presence of heavy metals in the soil. I tested this by analyzing the literature in regards to heavy metal based inhibition or promotion. Specifically, I not only analyzed if it is inhibition or promotion, but I also examined the extent and mechanisms by which these specific changes occur. My hypothesis supposed that heavy metals will promote antibiotic resistance and antibiotic production in soil bacteria and was tested in the following ways: 1) the effect of heavy metals, 2) the extent of the effect of heavy metals, and 3) the mechanisms by which these effects are undertaken. In regards to the results it was found that resistance was more prevalent than inhibition because of multiple factors including: presence of heavy metals in natural predatory mechanisms, greater antibiotic resistant genes in heavy metal environments, correlation between antibiotic resistance and heavy metal resistant genes, and plasmid amount in regards to environment metal concentration. While the results for inhibition, centered more on indirect factors like co-toxicity. In regards to the extent of resistance or inhibition I found that resistance data measured the extent to which a metal or metals confer resistance by looking at the number and type of antibiotics to which it confers resistance, while inhibition data was based off correlations between antibiotic resistance and heavy metals. Lastly in regards to mechanisms, I found that resistance mechanisms were centered on efflux systems, sequestration, and reduction while the research on inhibition mechanism moreover focused on biochemical pathways, interaction with antibiotics, and co-toxicity.
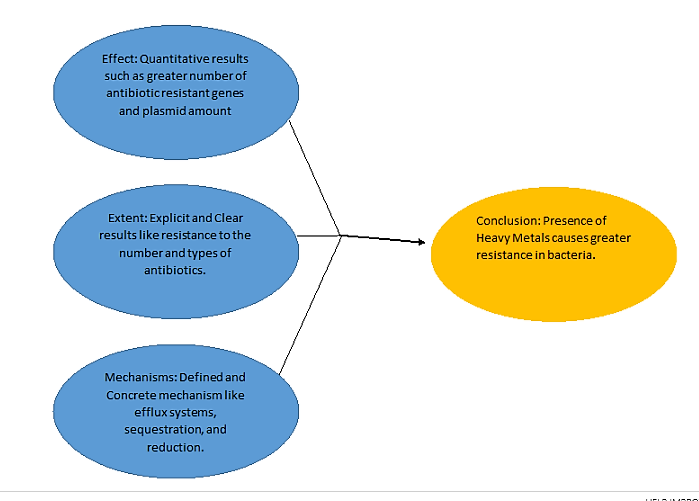
In summation, I found that the evidence went far and beyond to support the argument for resistance as compared to inhibition in the presence of heavy metals. In regards to the effect of heavy metals greater evidence was found for resistance than inhibition because the results for inhibition focused more on indirect factors like co-toxicity while the results on resistance were much more direct and quantitative. Secondly, I came to realize that the extent of metal based resistance was greater than the extent of inhibition because resistance data was better quality than inhibition data. The resistance data was clear and discreet based off the number and types of antibiotics to which it confers resistance, while inhibition data was based off weak correlations between antibiotic resistance and heavy metals. This trend was also prevalent in the mechanisms of resistance; the mechanisms for resistance were far more defined and concrete than the mechanisms for inhibition.. Thus, by looking at all these factors together I found that a stronger argument can be made for a rise in heavy metal based resistance (Figure 5).
Data courtesy of “Bacterial Multidrug Efflux Pumps: Much More Than Antibiotic Resistance Determinants ” (Blanco et al., 2016).
Although realizing that heavy metals promote resistance, I think it is more important to learn the mechanims by which they confer this resistance as that will help us realize the true essence of heavy metal based resistance. In summary, after looking through the literature on heavy metal based resistance, I found that three of the more critical mechanisms of resistance would include efflux systems, sequesteration, and reduction.
Mechanisms of Heavy Metal based inhibition of bacterial antimicrobial production and resistance
On the contrary, it is important to determine not the only the degree of decreased resistance but it is very important to acknowledge the role of mechanisms by which this occurs. Overall there are three very important mechanisms by which prokaryotes confer decreased resistance.
The first mechanism would be heavy metal based interactions with biochemical sites. In essence, metals like Cd, Hg, and Pb have a quite strong affinity for certain ligands like phosphates, histidyl, and cysteinyl side chains of proteins. By being able to act on such a wide variety of biochemical sites, these metals can inhibit a large variety of enzymes which may include antibiotic degrading enzymes (Vallee & Ulmer, 1972). Another study found that metals like copper can generate hydroxyl radicals while shifting from the reduced to the oxidized state, and these hydroxyl radicals can lead to nucleic acid, protein, and lipid damage (Festa & Thiele, 2011). Moreover, while looking at a study on the prokaryotic topoisomerase, gyrase, it was found that in the presence of calcium ions, gyrase was unable to conduct DNA cleavage at the rate of an enzyme that was not altered by the presence of calcium ions (Sissi & Palumbo, 2009). Clearly, metal ions play a very significant role in their alteration of biochemical pathways via their interaction with certain enzymes or other biomolecules.
The second mechanism would be heavy metal based interactions with antibiotics. Heavy metals can increase the bacteria is exposure to antibiotics by allowing certain proteins to bind them easier. In a study looking at S. aureus mercury and how it localized penicillin binding proteins which would thus allow certain antibiotics like penicillin to be able to bind it in an easier fashion (Paul et al., 1995). Thus, by letting antibiotics bind in an easier fashion, there is a decrease in resistance. In a study on Caulobacter crescentus, the protein StpX, a stalk-specific protein, responded to the presence of copper and zinc and this allowed for the identification and presence of penicillin-binding protein PbpC (Hughes et al., 2013). Therefore, by having metals that localize certain proteins that themselves localize antibiotic binding proteins or are antibiotic binding proteins, we can increase the effectivity of the antibiotic because it binds better resulting in better exposure and a decrease in overall resistance.
Lastly, this decrease in resistance could because of the co-toxic effects. This could mean that a heavy metal happens to degrade or inhibit a protein for antibiotic resistance that bacteria tend to use. Clearly, heavy metals play a large role in biochemical pathways, and have been shown to interact with certain ligands (Vallee & Ulmer, 1972). Moreover, byproducts of certain metal metabolism pathways may also interfere with proteins (Festa & Thiele, 2011). For example, mercury-inhibited enzymes might not regenerate after antibiotic based inhibition of protein synthesis (Holzel et al., 2012). By understanding these commonalities, I think we can understand how a decrease in resistance takes place because of co-toxic effects.
Although realizing that heavy metal mitigate resistance, I think it is more important to learn the mechanims by which they confer this decrease in resistance as that will help us realize the true essence of heavy metal inhibited resistance. In summary, after looking through the literature on heavy metal based resistance, I found that three of the more critical mechanisms of decreased resistance would include heavy metal interference with biochemical pathways, interaction with antibiotics, and co-toxicity.
Conclusion
The objective of this study was to engage in an extensive study on the antibiotic resistance or inhibition in regards in the presence of heavy metals. Specifically, my research question for this review was to examine if there is greater antibiotic resistance and production in soil bacteria because of the presence of heavy metals in the soil. I tested this by analyzing the literature in regards to heavy metal based inhibition or promotion. Specifically, I not only analyzed if it is inhibition or promotion, but I also examined the extent and mechanisms by which these specific changes occur. My hypothesis supposed that heavy metals will promote antibiotic resistance and antibiotic production in soil bacteria and was tested in the following ways: 1) the effect of heavy metals, 2) the extent of the effect of heavy metals, and 3) the mechanisms by which these effects are undertaken. In regards to the results it was found that resistance was more prevalent than inhibition because of multiple factors including: presence of heavy metals in natural predatory mechanisms, greater antibiotic resistant genes in heavy metal environments, correlation between antibiotic resistance and heavy metal resistant genes, and plasmid amount in regards to environment metal concentration. While the results for inhibition, centered more on indirect factors like co-toxicity. In regards to the extent of resistance or inhibition I found that resistance data measured the extent to which a metal or metals confer resistance by looking at the number and type of antibiotics to which it confers resistance, while inhibition data was based off correlations between antibiotic resistance and heavy metals. Lastly in regards to mechanisms, I found that resistance mechanisms were centered on efflux systems, sequestration, and reduction while the research on inhibition mechanism moreover focused on biochemical pathways, interaction with antibiotics, and co-toxicity.
In summation, I found that the evidence went far and beyond to support the argument for resistance as compared to inhibition in the presence of heavy metals. In regards to the effect of heavy metals greater evidence was found for resistance than inhibition because the results for inhibition focused more on indirect factors like co-toxicity while the results on resistance were much more direct and quantitative. Secondly, I came to realize that the extent of metal based resistance was greater than the extent of inhibition because resistance data was better quality than inhibition data. The resistance data was clear and discreet based off the number and types of antibiotics to which it confers resistance, while inhibition data was based off weak correlations between antibiotic resistance and heavy metals. This trend was also prevalent in the mechanisms of resistance; the mechanisms for resistance were far more defined and concrete than the mechanisms for inhibition.. Thus, by looking at all these factors together I found that a stronger argument can be made for a rise in heavy metal based resistance (Figure 5).
Figure 5: Causes of Greater Resistance in the Presence of Heavy Metals. This is a Schematic which depicts the three main causes of greater resistance in the presence of heavy metals.
Having realized this is very important because often we ignore heavy metal based waste products or runoffs and their pollution of our environments may have long term effects on the development of microbial resistance in those environments. Moreover, in a day age where microbial resistance is growing extremely rapidly we need to take a moreover cautious approach in how we micromanage microbial environments as how we manage our resources today could have disastrous long-term effects. Clear the results as a whole support the current thinking in the field where heavy metal presence promotes great resistance in bacteria. Ultimately this article serves to further the field’s thinking because it shows that although the evidence for resistance is stronger than the evidence for inhibition an argument can potentially be made for inhibition if we look at my sources or run more experiments.
Bibliography
Adegoke, A. A., Faleye, A. C., Singh, G., & Stenstrom, T. A. (2016). Antibiotic Resistant Superbugs: Assessment of the Interrelationship of Occurrence in Clinical Settings and Environmental Niches. Molecules, 22(1). doi:10.3390/molecules22010029
Alonso, A., Sanchez, P., & Martinez, J. L. (2001). Environmental selection of antibiotic resistance genes. Environ Microbiol, 3(1), 1-9.
Alvarez-Ortega, C., Olivares, J., & Martínez, J. L. (2013). RND multidrug efflux pumps: what are they good for? Front Microbiol, 4. doi:10.3389/fmicb.2013.00007
Amin, A., Latif, Z., & Opella, S. J. (2017). Expression and Purification of Transmembrane Protein MerE from Mercury Resistant Bacillus cereus. J Microbiol Biotechnol.doi:10.4014/jmb.1704.04062
Baker-Austin, C., Wright, M. S., Stepanauskas, R., & McArthur, J. V. (2006). Co-selection of antibiotic and metal resistance. Trends Microbiol, 14(4), 176-182. doi:10.1016/j.tim.2006.02.006
Barkay, T., Miller, S. M., & Summers, A. O. (2003). Bacterial mercury resistance from atoms to ecosystems. FEMS Microbiol Rev, 27(2-3), 355-384.
Blanco, P., Hernando-Amado, S., Reales-Calderon, J. A., Corona, F., Lira, F., Alcalde-Rico, M., . . . Martinez, J. L. (2016). Bacterial Multidrug Efflux Pumps: Much More Than Antibiotic Resistance Determinants. Microorganisms, 4(1). doi:10.3390/microorganisms4010014
Bontidean, I., Lloyd, J. R., Hobman, J. L., Wilson, J. R., Csoregi, E., Mattiasson, B., & Brown, N. L. (2000). Bacterial metal-resistance proteins and their use in biosensors for the detection of bioavailable heavy metals. J Inorg Biochem, 79(1-4), 225-229.
Brussow, H. (2017). Infection therapy: the problem of drug resistance - and possible solutions. Microb Biotechnol. doi:10.1111/1751-7915.12777
Chandrangsu, P., Rensing, C., & Helmann, J. D. (2017). Metal homeostasis and resistance in bacteria. Nat Rev Microbiol, 15(6), 338-350. doi:10.1038/nrmicro.2017.15
Chen, S., Li, X., Sun, G., Zhang, Y., Su, J., & Ye, J. (2015). Heavy Metal Induced Antibiotic Resistance in Bacterium LSJC7. Int J Mol Sci, 16(10), 23390-23404. doi:10.3390/ijms161023390
Cho, H., Jeong, D. W., Liu, Q., Yeo, W. S., Vogl, T., Skaar, E. P., . . . Bae, T. (2015). Calprotectin Increases the Activity of the SaeRS Two Component System and Murine Mortality during Staphylococcus aureus Infections. PLoS Pathog, 11(7), e1005026. doi:10.1371/journal.ppat.1005026
Davison, J. (1999). Genetic exchange between bacteria in the environment. Plasmid, 42(2), 73-91. doi:10.1006/plas.1999.1421
Delmar, J. A., Su, C. C., & Yu, E. W. (2014). Bacterial multidrug efflux transporters. Annu Rev Biophys, 43, 93-117. doi:10.1146/annurev-biophys-051013-022855
Diekema, D. J., BootsMiller, B. J., Vaughn, T. E., Woolson, R. F., Yankey, J. W., Ernst, E. J., . . . Doebbeling, B. N. (2004). Antimicrobial resistance trends and outbreak frequency in United States hospitals. Clin Infect Dis, 38(1), 78-85. doi:10.1086/380457
Festa, R. A., & Thiele, D. J. (2011). Copper: an essential metal in biology. Curr Biol, 21(21), R877-883. doi:10.1016/j.cub.2011.09.040
Fischbach, M. A., & Walsh, C. T. (2009). Antibiotics for emerging pathogens. Science, 325(5944), 1089-1093. doi:10.1126/science.1176667
Fisher, K., & Phillips, C. (2009). The ecology, epidemiology and virulence of Enterococcus. Microbiology, 155(Pt 6), 1749-1757. doi:10.1099/mic.0.026385-0
German, N., Doyscher, D., & Rensing, C. (2013). Bacterial killing in macrophages and amoeba: do they all use a brass dagger? Future Microbiol, 8(10), 1257-1264. doi:10.2217/fmb.13.100
Gross, M. (2013). Antibiotics in crisis. Curr Biol, 23(24), R1063-1065.
Handzlik, J., Matys, A., & Kiec-Kononowicz, K. (2013). Recent Advances in Multi-Drug Resistance (MDR) Efflux Pump Inhibitors of Gram-Positive Bacteria S. aureus. Antibiotics (Basel), 2(1), 28-45. doi:10.3390/antibiotics2010028
Hao, X., Luthje, F., Ronn, R., German, N. A., Li, X., Huang, F., . . . Rensing, C. (2016). A role for copper in protozoan grazing - two billion years selecting for bacterial copper resistance. Mol Microbiol, 102(4), 628-641. doi:10.1111/mmi.13483
Harrison, J. J., Ceri, H., & Turner, R. J. (2007). Multimetal resistance and tolerance in microbial biofilms. Nat Rev Microbiol, 5(12), 928-938. doi:10.1038/nrmicro1774
Harrison, J. J., Tremaroli, V., Stan, M. A., Chan, C. S., Vacchi-Suzzi, C., Heyne, B. J., . . . Turner, R. J. (2009). Chromosomal antioxidant genes have metal ion-specific roles as determinants of bacterial metal tolerance. Environ Microbiol, 11(10), 2491-2509. doi:10.1111/j.1462-2920.2009.01973.x
Hobman, J. L., & Crossman, L. C. (2015). Bacterial antimicrobial metal ion resistance. J Med Microbiol, 64(Pt 5), 471-497. doi:10.1099/jmm.0.023036-0
Holzel, C. S., Muller, C., Harms, K. S., Mikolajewski, S., Schafer, S., Schwaiger, K., & Bauer, J. (2012). Heavy metals in liquid pig manure in light of bacterial antimicrobial resistance. Environ Res, 113, 21-27. doi:10.1016/j.envres.2012.01.002
Hu, Y., Cheng, H., & Tao, S. (2017). Environmental and human health challenges of industrial livestock and poultry farming in China and their mitigation. Environ Int, 107, 111-130. doi:10.1016/j.envint.2017.07.003
Hughes, H. V., Lisher, J. P., Hardy, G. G., Kysela, D. T., Arnold, R. J., Giedroc, D. P., & Brun, Y. V. (2013). Coordinate synthesis and protein localization in a bacterial organelle by the action of a penicillin-binding-protein. Molecular microbiology, 90(6), 10.1111/mmi.12422. doi:10.1111/mmi.12422
Lima de Silva, A. A., de Carvalho, M. A., de Souza, S. A., Dias, P. M., da Silva Filho, R. G., de Meirelles Saramago, C. S., . . . Hofer, E. (2012). Heavy metal tolerance (Cr, Ag AND Hg) in bacteria isolated from sewage. Braz J Microbiol, 43(4), 1620-1631. doi:10.1590/S1517-838220120004000047
Lin, H., Sun, W., Zhang, Z., Chapman, S. J., Freitag, T. E., Fu, J., . . . Ma, J. (2016). Effects of manure and mineral fertilization strategies on soil antibiotic resistance gene levels and microbial community in a paddy-upland rotation system. Environ Pollut, 211, 332-337. doi:10.1016/j.envpol.2016.01.007
Lovley, D. R., & Phillips, E. J. (1988). Novel mode of microbial energy metabolism: organic carbon oxidation coupled to dissimilatory reduction of iron or manganese. Appl Environ Microbiol, 54(6), 1472-1480.
Lu, M. (2016). Structures of multidrug and toxic compound extrusion transporters and their mechanistic implications. Channels (Austin), 10(2), 88-100. doi:10.1080/19336950.2015.1106654
Lubelski, J., Konings, W. N., & Driessen, A. J. M. (2007). Distribution and Physiology of ABC-Type Transporters Contributing to Multidrug Resistance in Bacteria. Microbiol Mol Biol Rev, 71(3), 463-476. doi:10.1128/mmbr.00001-07
McHugh, G. L., Moellering, R. C., Hopkins, C. C., & Swartz, M. N. (1975). Salmonella typhimurium resistant to silver nitrate, chloramphenicol, and ampicillin. Lancet, 1(7901), 235-240.
Mergeay, M., Nies, D., Schlegel, H. G., Gerits, J., Charles, P., & Van Gijsegem, F. (1985). Alcaligenes eutrophus CH34 is a facultative chemolithotroph with plasmid- bound resistance to heavy metals. J Bacteriol, 162(1), 328-334.
Mukhopadhyay, R., & Rosen, B. P. (2002). Arsenate reductases in prokaryotes and eukaryotes. Environ Health Perspect, 110 Suppl 5, 745-748.
Nicholson, F. A., Smith, S. R., Alloway, B. J., Carlton-Smith, C., & Chambers, B. J. (2003). An inventory of heavy metals inputs to agricultural soils in England and Wales. Sci Total Environ, 311(1-3), 205-219. doi:10.1016/S0048-9697(03)00139-6
Nies, D. H. (1999). Microbial heavy-metal resistance. Appl Microbiol Biotechnol, 51(6), 730-750.
Nies, D. H. (2003). Efflux-mediated heavy metal resistance in prokaryotes. FEMS Microbiol Rev, 27(2-3), 313-339.
Norman, G., Christie, J., Liu, Z., Westby, M. J., Jefferies, J. M., Hudson, T., . . . Dumville, J. C. (2017). Antiseptics for burns. Cochrane Database Syst Rev, 7, CD011821. doi:10.1002/14651858.CD011821.pub2
Pao, S. S., Paulsen, I. T., & Saier, M. H. (1998). Major Facilitator Superfamily. Microbiol Mol Biol Rev, 62(1), 1-34.
Paul, T. R., Venter, A., Blaszczak, L. C., Parr, T. R., Jr., Labischinski, H., & Beveridge, T. J. (1995). Localization of penicillin-binding proteins to the splitting system of Staphylococcus aureus septa by using a mercury-penicillin V derivative. J Bacteriol, 177(13), 3631-3640.
Poole, K. (2017). At the Nexus of Antibiotics and Metals: The Impact of Cu and Zn on Antibiotic Activity and Resistance. Trends Microbiol. doi:10.1016/j.tim.2017.04.010
Porcheron, G., Garenaux, A., Proulx, J., Sabri, M., & Dozois, C. M. (2013). Iron, copper, zinc, and manganese transport and regulation in pathogenic Enterobacteria: correlations between strains, site of infection and the relative importance of the different metal transport systems for virulence. Front Cell Infect Microbiol, 3, 90. doi:10.3389/fcimb.2013.00090
Pruitt, B. A., Jr., McManus, A. T., Kim, S. H., & Goodwin, C. W. (1998). Burn wound infections: current status. World J Surg, 22(2), 135-145.
Rasmussen, L. D., & Sorensen, S. J. (1998). The effect of longterm exposure to mercury on the bacterial community in marine sediment. Curr Microbiol, 36(5), 291- 297.
Schiering, N., Kabsch, W., Moore, M. J., Distefano, M. D., Walsh, C. T., & Pai, E. F. (1991). Structure of the detoxification catalyst mercuric ion reductase from Bacillus sp. strain RC607. Nature, 352(6331), 168-172. doi:10.1038/352168a0
Schulz-Zunkel, C., & Krueger, F. (2009). Trace metal dynamics in floodplain soils of the river Elbe: a review. J Environ Qual, 38(4), 1349-1362. doi:10.2134/jeq2008.0299
Seiler, C., & Berendonk, T. U. (2012). Heavy metal driven co-selection of antibiotic resistance in soil and water bodies impacted by agriculture and aquaculture. Front Microbiol, 3, 399. doi:10.3389/fmicb.2012.00399
Silver, S., & Phung, L. T. (1996). Bacterial heavy metal resistance: new surprises. Annu Rev Microbiol, 50, 753-789. doi:10.1146/annurev.micro.50.1.753
Sissi, C., & Palumbo, M. (2009). Effects of magnesium and related divalent metal ions in topoisomerase structure and function. Nucleic Acids Res, 37(3), 702-711. doi:10.1093/nar/gkp024
Stepanauskas, R., Glenn, T. C., Jagoe, C. H., Tuckfield, R. C., Lindell, A. H., & McArthur, J. V. (2005). Elevated microbial tolerance to metals and antibiotics in metal- contaminated industrial environments. Environ Sci Technol, 39(10), 3671-3678.
Sterritt, R. M., & Lester, J. N. (1980). Interactions of heavy metals with bacteria. Sci Total Environ, 14(1), 5-17.
Tuckfield, R. C., & McArthur, J. V. (2008). Spatial analysis of antibiotic resistance along metal contaminated streams. Microb Ecol, 55(4), 595-607. doi:10.1007/s00248-007-9303-5
Vallee, B. L., & Ulmer, D. D. (1972). Biochemical effects of mercury, cadmium, and lead. Annu Rev Biochem, 41(10), 91-128. doi:10.1146/annurev.bi.41.070172.000515
Ventola, C. L. (2015). The antibiotic resistance crisis: part 1: causes and threats. P T, 40(4), 277-283.
Viswanathan, V. K. (2014). Off-label abuse of antibiotics by bacteria. Gut Microbes, 5(1), 3-4. doi:10.4161/gmic.28027
Yousefi, M., Dadashpour, M., Hejazi, M., Hasanzadeh, M., Behnam, B., de la Guardia, M., . . . Mokhtarzadeh, A. (2017). Anti-bacterial activity of graphene oxide as a new weapon nanomaterial to combat multidrug-resistance bacteria. Mater Sci Eng C Mater Biol Appl, 74, 568-581. doi:10.1016/j.msec.2016.12.125
Zhu, Y. G., Johnson, T. A., Su, J. Q., Qiao, M., Guo, G. X., Stedtfeld, R. D., . . . Tiedje, J. M. (2013). Diverse and abundant antibiotic resistance genes in Chinese swine farms. Proc Natl Acad Sci U S A, 110(9), 3435-3440. doi:10.1073/pnas.1222743110
Zinner, S. H. (2005). The search for new antimicrobials: why we need new options. Expert Rev Anti Infect Ther, 3(6), 907-913. doi:10.1586/14787210.3.6.907
Having realized this is very important because often we ignore heavy metal based waste products or runoffs and their pollution of our environments may have long term effects on the development of microbial resistance in those environments. Moreover, in a day age where microbial resistance is growing extremely rapidly we need to take a moreover cautious approach in how we micromanage microbial environments as how we manage our resources today could have disastrous long-term effects. Clear the results as a whole support the current thinking in the field where heavy metal presence promotes great resistance in bacteria. Ultimately this article serves to further the field’s thinking because it shows that although the evidence for resistance is stronger than the evidence for inhibition an argument can potentially be made for inhibition if we look at my sources or run more experiments.
Bibliography
Adegoke, A. A., Faleye, A. C., Singh, G., & Stenstrom, T. A. (2016). Antibiotic Resistant Superbugs: Assessment of the Interrelationship of Occurrence in Clinical Settings and Environmental Niches. Molecules, 22(1). doi:10.3390/molecules22010029
Alonso, A., Sanchez, P., & Martinez, J. L. (2001). Environmental selection of antibiotic resistance genes. Environ Microbiol, 3(1), 1-9.
Alvarez-Ortega, C., Olivares, J., & Martínez, J. L. (2013). RND multidrug efflux pumps: what are they good for? Front Microbiol, 4. doi:10.3389/fmicb.2013.00007
Amin, A., Latif, Z., & Opella, S. J. (2017). Expression and Purification of Transmembrane Protein MerE from Mercury Resistant Bacillus cereus. J Microbiol Biotechnol.doi:10.4014/jmb.1704.04062
Baker-Austin, C., Wright, M. S., Stepanauskas, R., & McArthur, J. V. (2006). Co-selection of antibiotic and metal resistance. Trends Microbiol, 14(4), 176-182. doi:10.1016/j.tim.2006.02.006
Barkay, T., Miller, S. M., & Summers, A. O. (2003). Bacterial mercury resistance from atoms to ecosystems. FEMS Microbiol Rev, 27(2-3), 355-384.
Blanco, P., Hernando-Amado, S., Reales-Calderon, J. A., Corona, F., Lira, F., Alcalde-Rico, M., . . . Martinez, J. L. (2016). Bacterial Multidrug Efflux Pumps: Much More Than Antibiotic Resistance Determinants. Microorganisms, 4(1). doi:10.3390/microorganisms4010014
Bontidean, I., Lloyd, J. R., Hobman, J. L., Wilson, J. R., Csoregi, E., Mattiasson, B., & Brown, N. L. (2000). Bacterial metal-resistance proteins and their use in biosensors for the detection of bioavailable heavy metals. J Inorg Biochem, 79(1-4), 225-229.
Brussow, H. (2017). Infection therapy: the problem of drug resistance - and possible solutions. Microb Biotechnol. doi:10.1111/1751-7915.12777
Chandrangsu, P., Rensing, C., & Helmann, J. D. (2017). Metal homeostasis and resistance in bacteria. Nat Rev Microbiol, 15(6), 338-350. doi:10.1038/nrmicro.2017.15
Chen, S., Li, X., Sun, G., Zhang, Y., Su, J., & Ye, J. (2015). Heavy Metal Induced Antibiotic Resistance in Bacterium LSJC7. Int J Mol Sci, 16(10), 23390-23404. doi:10.3390/ijms161023390
Cho, H., Jeong, D. W., Liu, Q., Yeo, W. S., Vogl, T., Skaar, E. P., . . . Bae, T. (2015). Calprotectin Increases the Activity of the SaeRS Two Component System and Murine Mortality during Staphylococcus aureus Infections. PLoS Pathog, 11(7), e1005026. doi:10.1371/journal.ppat.1005026
Davison, J. (1999). Genetic exchange between bacteria in the environment. Plasmid, 42(2), 73-91. doi:10.1006/plas.1999.1421
Delmar, J. A., Su, C. C., & Yu, E. W. (2014). Bacterial multidrug efflux transporters. Annu Rev Biophys, 43, 93-117. doi:10.1146/annurev-biophys-051013-022855
Diekema, D. J., BootsMiller, B. J., Vaughn, T. E., Woolson, R. F., Yankey, J. W., Ernst, E. J., . . . Doebbeling, B. N. (2004). Antimicrobial resistance trends and outbreak frequency in United States hospitals. Clin Infect Dis, 38(1), 78-85. doi:10.1086/380457
Festa, R. A., & Thiele, D. J. (2011). Copper: an essential metal in biology. Curr Biol, 21(21), R877-883. doi:10.1016/j.cub.2011.09.040
Fischbach, M. A., & Walsh, C. T. (2009). Antibiotics for emerging pathogens. Science, 325(5944), 1089-1093. doi:10.1126/science.1176667
Fisher, K., & Phillips, C. (2009). The ecology, epidemiology and virulence of Enterococcus. Microbiology, 155(Pt 6), 1749-1757. doi:10.1099/mic.0.026385-0
German, N., Doyscher, D., & Rensing, C. (2013). Bacterial killing in macrophages and amoeba: do they all use a brass dagger? Future Microbiol, 8(10), 1257-1264. doi:10.2217/fmb.13.100
Gross, M. (2013). Antibiotics in crisis. Curr Biol, 23(24), R1063-1065.
Handzlik, J., Matys, A., & Kiec-Kononowicz, K. (2013). Recent Advances in Multi-Drug Resistance (MDR) Efflux Pump Inhibitors of Gram-Positive Bacteria S. aureus. Antibiotics (Basel), 2(1), 28-45. doi:10.3390/antibiotics2010028
Hao, X., Luthje, F., Ronn, R., German, N. A., Li, X., Huang, F., . . . Rensing, C. (2016). A role for copper in protozoan grazing - two billion years selecting for bacterial copper resistance. Mol Microbiol, 102(4), 628-641. doi:10.1111/mmi.13483
Harrison, J. J., Ceri, H., & Turner, R. J. (2007). Multimetal resistance and tolerance in microbial biofilms. Nat Rev Microbiol, 5(12), 928-938. doi:10.1038/nrmicro1774
Harrison, J. J., Tremaroli, V., Stan, M. A., Chan, C. S., Vacchi-Suzzi, C., Heyne, B. J., . . . Turner, R. J. (2009). Chromosomal antioxidant genes have metal ion-specific roles as determinants of bacterial metal tolerance. Environ Microbiol, 11(10), 2491-2509. doi:10.1111/j.1462-2920.2009.01973.x
Hobman, J. L., & Crossman, L. C. (2015). Bacterial antimicrobial metal ion resistance. J Med Microbiol, 64(Pt 5), 471-497. doi:10.1099/jmm.0.023036-0
Holzel, C. S., Muller, C., Harms, K. S., Mikolajewski, S., Schafer, S., Schwaiger, K., & Bauer, J. (2012). Heavy metals in liquid pig manure in light of bacterial antimicrobial resistance. Environ Res, 113, 21-27. doi:10.1016/j.envres.2012.01.002
Hu, Y., Cheng, H., & Tao, S. (2017). Environmental and human health challenges of industrial livestock and poultry farming in China and their mitigation. Environ Int, 107, 111-130. doi:10.1016/j.envint.2017.07.003
Hughes, H. V., Lisher, J. P., Hardy, G. G., Kysela, D. T., Arnold, R. J., Giedroc, D. P., & Brun, Y. V. (2013). Coordinate synthesis and protein localization in a bacterial organelle by the action of a penicillin-binding-protein. Molecular microbiology, 90(6), 10.1111/mmi.12422. doi:10.1111/mmi.12422
Lima de Silva, A. A., de Carvalho, M. A., de Souza, S. A., Dias, P. M., da Silva Filho, R. G., de Meirelles Saramago, C. S., . . . Hofer, E. (2012). Heavy metal tolerance (Cr, Ag AND Hg) in bacteria isolated from sewage. Braz J Microbiol, 43(4), 1620-1631. doi:10.1590/S1517-838220120004000047
Lin, H., Sun, W., Zhang, Z., Chapman, S. J., Freitag, T. E., Fu, J., . . . Ma, J. (2016). Effects of manure and mineral fertilization strategies on soil antibiotic resistance gene levels and microbial community in a paddy-upland rotation system. Environ Pollut, 211, 332-337. doi:10.1016/j.envpol.2016.01.007
Lovley, D. R., & Phillips, E. J. (1988). Novel mode of microbial energy metabolism: organic carbon oxidation coupled to dissimilatory reduction of iron or manganese. Appl Environ Microbiol, 54(6), 1472-1480.
Lu, M. (2016). Structures of multidrug and toxic compound extrusion transporters and their mechanistic implications. Channels (Austin), 10(2), 88-100. doi:10.1080/19336950.2015.1106654
Lubelski, J., Konings, W. N., & Driessen, A. J. M. (2007). Distribution and Physiology of ABC-Type Transporters Contributing to Multidrug Resistance in Bacteria. Microbiol Mol Biol Rev, 71(3), 463-476. doi:10.1128/mmbr.00001-07
McHugh, G. L., Moellering, R. C., Hopkins, C. C., & Swartz, M. N. (1975). Salmonella typhimurium resistant to silver nitrate, chloramphenicol, and ampicillin. Lancet, 1(7901), 235-240.
Mergeay, M., Nies, D., Schlegel, H. G., Gerits, J., Charles, P., & Van Gijsegem, F. (1985). Alcaligenes eutrophus CH34 is a facultative chemolithotroph with plasmid- bound resistance to heavy metals. J Bacteriol, 162(1), 328-334.
Mukhopadhyay, R., & Rosen, B. P. (2002). Arsenate reductases in prokaryotes and eukaryotes. Environ Health Perspect, 110 Suppl 5, 745-748.
Nicholson, F. A., Smith, S. R., Alloway, B. J., Carlton-Smith, C., & Chambers, B. J. (2003). An inventory of heavy metals inputs to agricultural soils in England and Wales. Sci Total Environ, 311(1-3), 205-219. doi:10.1016/S0048-9697(03)00139-6
Nies, D. H. (1999). Microbial heavy-metal resistance. Appl Microbiol Biotechnol, 51(6), 730-750.
Nies, D. H. (2003). Efflux-mediated heavy metal resistance in prokaryotes. FEMS Microbiol Rev, 27(2-3), 313-339.
Norman, G., Christie, J., Liu, Z., Westby, M. J., Jefferies, J. M., Hudson, T., . . . Dumville, J. C. (2017). Antiseptics for burns. Cochrane Database Syst Rev, 7, CD011821. doi:10.1002/14651858.CD011821.pub2
Pao, S. S., Paulsen, I. T., & Saier, M. H. (1998). Major Facilitator Superfamily. Microbiol Mol Biol Rev, 62(1), 1-34.
Paul, T. R., Venter, A., Blaszczak, L. C., Parr, T. R., Jr., Labischinski, H., & Beveridge, T. J. (1995). Localization of penicillin-binding proteins to the splitting system of Staphylococcus aureus septa by using a mercury-penicillin V derivative. J Bacteriol, 177(13), 3631-3640.
Poole, K. (2017). At the Nexus of Antibiotics and Metals: The Impact of Cu and Zn on Antibiotic Activity and Resistance. Trends Microbiol. doi:10.1016/j.tim.2017.04.010
Porcheron, G., Garenaux, A., Proulx, J., Sabri, M., & Dozois, C. M. (2013). Iron, copper, zinc, and manganese transport and regulation in pathogenic Enterobacteria: correlations between strains, site of infection and the relative importance of the different metal transport systems for virulence. Front Cell Infect Microbiol, 3, 90. doi:10.3389/fcimb.2013.00090
Pruitt, B. A., Jr., McManus, A. T., Kim, S. H., & Goodwin, C. W. (1998). Burn wound infections: current status. World J Surg, 22(2), 135-145.
Rasmussen, L. D., & Sorensen, S. J. (1998). The effect of longterm exposure to mercury on the bacterial community in marine sediment. Curr Microbiol, 36(5), 291- 297.
Schiering, N., Kabsch, W., Moore, M. J., Distefano, M. D., Walsh, C. T., & Pai, E. F. (1991). Structure of the detoxification catalyst mercuric ion reductase from Bacillus sp. strain RC607. Nature, 352(6331), 168-172. doi:10.1038/352168a0
Schulz-Zunkel, C., & Krueger, F. (2009). Trace metal dynamics in floodplain soils of the river Elbe: a review. J Environ Qual, 38(4), 1349-1362. doi:10.2134/jeq2008.0299
Seiler, C., & Berendonk, T. U. (2012). Heavy metal driven co-selection of antibiotic resistance in soil and water bodies impacted by agriculture and aquaculture. Front Microbiol, 3, 399. doi:10.3389/fmicb.2012.00399
Silver, S., & Phung, L. T. (1996). Bacterial heavy metal resistance: new surprises. Annu Rev Microbiol, 50, 753-789. doi:10.1146/annurev.micro.50.1.753
Sissi, C., & Palumbo, M. (2009). Effects of magnesium and related divalent metal ions in topoisomerase structure and function. Nucleic Acids Res, 37(3), 702-711. doi:10.1093/nar/gkp024
Stepanauskas, R., Glenn, T. C., Jagoe, C. H., Tuckfield, R. C., Lindell, A. H., & McArthur, J. V. (2005). Elevated microbial tolerance to metals and antibiotics in metal- contaminated industrial environments. Environ Sci Technol, 39(10), 3671-3678.
Sterritt, R. M., & Lester, J. N. (1980). Interactions of heavy metals with bacteria. Sci Total Environ, 14(1), 5-17.
Tuckfield, R. C., & McArthur, J. V. (2008). Spatial analysis of antibiotic resistance along metal contaminated streams. Microb Ecol, 55(4), 595-607. doi:10.1007/s00248-007-9303-5
Vallee, B. L., & Ulmer, D. D. (1972). Biochemical effects of mercury, cadmium, and lead. Annu Rev Biochem, 41(10), 91-128. doi:10.1146/annurev.bi.41.070172.000515
Ventola, C. L. (2015). The antibiotic resistance crisis: part 1: causes and threats. P T, 40(4), 277-283.
Viswanathan, V. K. (2014). Off-label abuse of antibiotics by bacteria. Gut Microbes, 5(1), 3-4. doi:10.4161/gmic.28027
Yousefi, M., Dadashpour, M., Hejazi, M., Hasanzadeh, M., Behnam, B., de la Guardia, M., . . . Mokhtarzadeh, A. (2017). Anti-bacterial activity of graphene oxide as a new weapon nanomaterial to combat multidrug-resistance bacteria. Mater Sci Eng C Mater Biol Appl, 74, 568-581. doi:10.1016/j.msec.2016.12.125
Zhu, Y. G., Johnson, T. A., Su, J. Q., Qiao, M., Guo, G. X., Stedtfeld, R. D., . . . Tiedje, J. M. (2013). Diverse and abundant antibiotic resistance genes in Chinese swine farms. Proc Natl Acad Sci U S A, 110(9), 3435-3440. doi:10.1073/pnas.1222743110
Zinner, S. H. (2005). The search for new antimicrobials: why we need new options. Expert Rev Anti Infect Ther, 3(6), 907-913. doi:10.1586/14787210.3.6.907